Novo Nordisk shares in Copenhagen finally caught a bid after the U.S. Food and Drug Administration issued 30 warning letters to telehealth companies for making false and misleading claims regarding compounded GLP-1 products (otherwise known as copycat GLP-1s) offered on their websites.
FDA has warned 30 telehealth companies about misleading drug ads.
Consumers should not be fooled into thinking that unapproved, compounded GLP-1 drugs—marketed by telehealth companies—are generic versions or the same as FDA-approved products.
— Dr. Marty Makary (@DrMakaryFDA) March 3, 2026
Citi analyst Geoff Meacham told clients that a quick scan of some of the warning letters “shows the agency is taking issue with telehealth companies calling their compounded products’ generic Zepbound’ or ‘generic Mounjaro’ when these products are not FDA-approved.”
“It’s a new era. We are paying close attention to misleading claims being made by telehealth and pharma companies across all media platforms—and taking swift action,” FDA Commissioner Marty Makary wrote in a statement.
Makary noted, “Compounded drugs can be important for overcoming shortages or meeting unique patient needs—but compounders should not try to compound drugs in a way that circumvents FDA’s approval process.”
Novo and the telehealth firm Hims & Hers have been locked in a GLP-1 battle over the firm’s copycat GLP-1 drugs. Sagging demand, lower prices, and copycat GLP-1s have pressured Novo’s outlook for the year.
Hims & Hers
Novo Wegovy
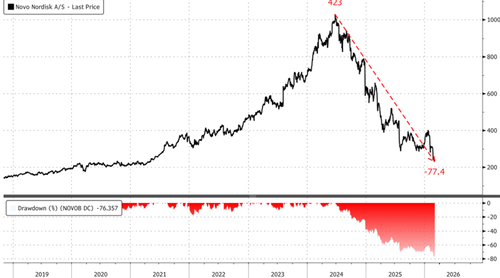
Shares of Novo caught a bid in Copenhagen, rising about 5%, but the key question is: who is stepping in to catch this falling knife?
The latest on Novo and the GLP-1 feud:
Meanwhile, Novo’s biggest bull, Goldman analyst James Quigley, downgraded the stock from “Buy” to “Hold” earlier this week. Quigley’s full note can be viewed here and is available to pro subs.
Loading recommendations…